providing caregivers the visibility they need to make safe, informed and precise clinical decisions in
realtime. Precision Ventilation is finally a reality at the bedside

By taking into account patient ́s lung heterogeneities and their unique physiological responses to mechanical ventilation, ENLIGHT makes Precision Medicine a reality for ventilated patients, protecting them from the hidden dangers and unwanted side effects caused by standard mechanical ventilation.

ENLIGHT is a bedside expert system that provides real-time facts freeing caregivers from the limitation of making decisions based on assumptions. By giving instantaneous visual feedback of each patient’s individual response to ventilatory interventions, caregivers can foster the most appropriate ventilation strategy right from the beginning, to improve patient’s outcomes.

ENLIGHT saves time at a moment when it counts most, giving early visibility to otherwise hidden problems at the precise time of their occurrence, long before their clinical manifestations are detected by routine examinations. Problems can be treated before they turn into more serious complications.
Intuitive: integrated clinical
decision support tools
Realtime: high temporal
resolution
Easy to install, simple to use
Transportable, can
benefit multiple patients
Comfortable,
ergonomic belts
Non-Invasive
Bedside: no
patient transport
Radiation Free - can be used frequently
Provides continuous,
real-time images
Operator independent
The Ventilation Map, Dynamic Image, Plethysmogram and advanced Ventilatory Parameters provide
instantaneous visual feedback of each patient’s individual response to interventions, fostering an
individualized, targeted and timely ventilation.
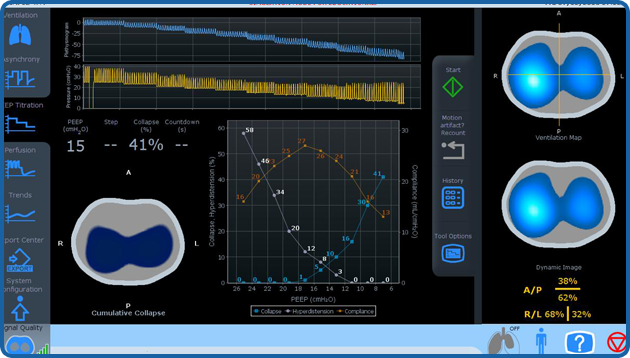
With the PEEP Titration Tool, caregivers can better cope with patient’s lungs heterogeneity, abnormal pleural pressures, and
the unique physiological response to a PEEP challenge. The detection of the individual best level of PEEP enables to minimize
driving pressure and collapse delivering a targeted precision ventilation.
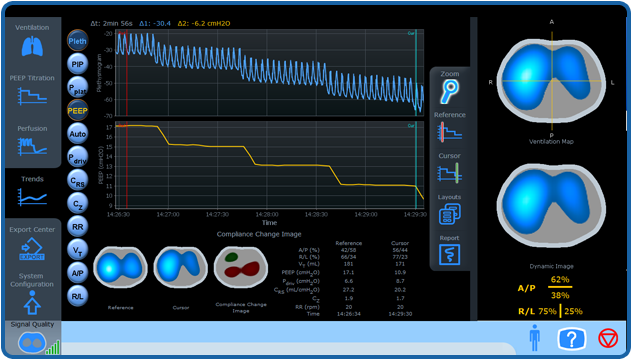
With the Trends Screen you can timely find out what went wrong and decide what to do next. Is an interactive tool that
allows visualization of Electrical Impedance Tomography data, ventilation parameters and pulmonary mechanics, making it
easier to detect events and interpret data intuitively.
Asynchronies are frequent but most of the time underdiagnosed, and are associated with worse clinical outcomes.
The Asynchrony Tool tracks the occurrence of Double and Reverse Trigger and quantifies the volume delivered during stacked cycles.
 The Technology
The TechnologyElectrical Impedance Tomography (EIT) makes use of high frequency, low intensity electric signals, to provide live images of the breathing lung showing pulmonary performance in real time.
The electrical signals are harmless to tissue and therefore suitable for long-term use, are transmitted and received through electrodes built into a specially designed belt positioned around the patient’s chest.
As different types of tissue have a different levels of impedance to the electrical signals, these differences can be used to generate images by using uniquely designed, clinically validated algorithm.
EIT is both non-invasive and radiation free ideally for long term, bedside applications.
Explore the LiteratureWe are passionate and moved by a cause: to make precision medicine a reality in ventilation to benefit patients, caregivers, hospitals and payers. With a highly qualified multidisciplinary team, we focus on the development of state-of-the-art ventilation!
We are disruptive, in constant evolution: We promote a strong partnership between academy and industry. Invested by FinHealth, a private equity and venture capital fund manager focused exclusively on health investments, we also received support from FAPESP and FINEP, brazilian public research companies.